By RENEE STOVSKY
April 24, 2020
Attorney Walter Lamkin, 69, considers Vail, Colorado his "spiritual home," and he usually spends at least a month there each winter, skiing, hiking and climbing.
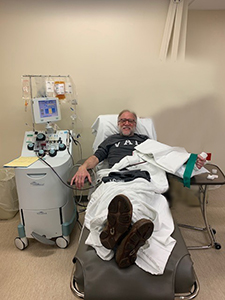
Having recovered from COVID-19, Walter Lamkin donates the first convalescent plasma at Mercy Hospital St. Louis.
This year was a no different, although Lamkin recalls running a fever one night in late February, followed by a lingering cough. He didn't think much of it at the time, and was back on the slopes the first week in March, volunteering as a mountain host, attending an annual luncheon to thank the ski patrol and hosting a group of friends for an annual party on the summit.
Two days after he flew home to St. Louis from his alpine retreat, on March 11, he received an e-mail from Eagle County, Colorado, informing him he may have been exposed to novel coronavirus. Initially he shrugged it off. But the next day he learned that one of his good pals, someone who had been with him "day and night" there, had been airlifted to Denver in a coma. "That got my attention," he says.
Lamkin obtained a COVID-19 test at a newly established Mercy drive-thru testing center and got the results on March 21. "I learned I was positive the same day my friend died of coronavirus in Denver," he says. "That was pretty rough."
Lamkin was released from quarantine on April 1, and because he is assumed to have some immunity to the disease, has become the de facto grocery shopper and delivery boy for family and friends under stay-at-home orders.
"Several more of my Colorado friends have since been sickened by COVID-19, and I've lost another person in my life, this one in Cincinnati, to the virus. So, I feel lucky to have the flexibility to help other people out," Lamkin says.
Pay it forward
Just three days after his quarantine ended, on April 4, Lamkin got a call from Dr. Emily Schindler, medical director of Mercy Blood Donor Services at Mercy Hospital St. Louis.

Dr. Emily Schindler, medical director of Mercy Blood Donor Services at Mercy Hospital St. Louis, is part of the team coordinating convalescent plasma treatments in the Mercy system.
"She asked me if I would consider donating plasma for COVID-19 patients who are critically ill, and I agreed without hesitation," says Lamkin. "I feel so fortunate to have had a mild case, and I want to help others with their recovery now."
After another test to confirm he was virus-free, Lamkin became the first such plasma donor at Mercy on April 6. Three days later, his 600-milliliter donation was transfused among three COVID-19 patients, all on ventilators in the ICU at Mercy Hospital St. Louis.
Though Schindler cautions that clinical trials are needed to determine the efficacy of convalescent plasma treatment, she says there are early clinical signs of improvement following treatment and doctors have been able to decrease patients' ventilator settings or remove patients from ventilator assisted respiration altogether in some cases.
"This is not an immediate fix; patients are still very sick" after the treatment, says Schindler, adding that patients are showing positive signs more rapidly than clinicians had originally hoped.
Meticulous coordination
Earlier this month, the Food and Drug Administration announced a national study to track the experimental use of convalescent plasma as well as hyperimmune globulin in COVID-19 patients meeting certain criteria. Because of its own blood donor program and strong relationships with community blood bank partners, Mercy and Mercy Research was well-positioned to participate in the trials.
"This is truly a team effort," says Schindler. "It takes 20-30 people to coordinate this, from the recruiting standpoint to the clinical oversight of apheresis and blood matching, to regulatory and IT support."
A research team drawn from within the Mercy system is matching products for all 12 Mercy hospitals in the state of Missouri, as well as connecting hospitals across its system —including those in Arkansas, Kansas and Oklahoma — with other blood collection agencies.
Time-tested therapy
Though convalescent plasma has not yet been shown effective specifically in treating COVID-19, it is a therapy that has been used in medicine for more than a century, predating vaccines and antibiotics. Survivors of viral pandemics are thought to carry antibodies that successfully attacked the virus. Those antibodies sometimes can remain in their blood for years.
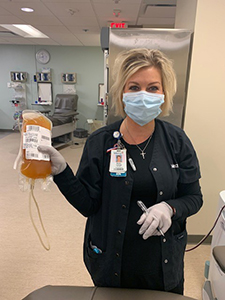
Robin Cornett, RN, clinical nurse with Mercy Blood Donor Services, carries a bag of plasm.
"In layman's terms, it's a way to try to transplant immune response from a recovered patient into a struggling patient," says Schindler.
During the Spanish flu pandemic of 1918, two physicians at a naval hospital in Massachusetts tried the treatment with enough success to warrant further testing. More recently, it has been used in the 2003 severe acute respiratory syndrome (SARS-CoV-1) epidemic, the 2009-2010 H1N1 influenza virus 9 (swine flu) pandemic, the 2012 Middle East respiratory syndrome (MERS-CoV) epidemic, and the 2014 Ebola virus disease (EVD) epidemic.
"This is an important area of research — the use of products made from a recovered patient's blood to potentially treat COVID-19," FDA commissioner Stephen Hahn said in a release announcing the trials. "The FDA had played a key role in organizing a partnership between industry, academic institutions and government agencies to facilitate expanded access to convalescent plasma. This is certainly a great example of how we can all come together to take swift action to help the American people during a crisis."
Vetting donors
Since Lamkin's initial plasma donation, Mercy has obtained convalescent plasma from 19 donors who have recovered from COVID-19. By midafternoon on April 23, 21 Mercy patients had been infused with the plasma.
At this point, it is still a laborious process to identify and certify donors. According to FDA guidelines, patients must have had a known positive test, completely recovered, been asymptomatic for 14 days and then have had a known negative test. Once eligible donors have given blood, those units must be tested for other infectious diseases and blood types must be matched with potential recipients. Patients need signed informed consents to participate, and must be eligible for emergency investigational new drug programs. Strict guidelines for labeling and record keeping must be followed by blood centers and hospitals and results reported back to the FDA.
Much is also unknown about possible side effects — from the regular risks of blood transfusions, such as allergic reactions and fluid overload — to more long-term conditions. And it is still unclear how often donors can give plasma; estimates range from every 28 days to every four months at this point.
Of course, convalescent plasma is not the only option health care professionals are trying during this pandemic — others include the antiviral treatment Remdesiver, used during the Ebola crisis, and antimalarial/immune suppressants chloroquine and hydroxychloroquine.
"As doctors, we are feeling pretty overwhelmed, with limited resources to fight this coronavirus. Plasma sounds promising, though at this point there are no quantified results," says Schindler.
"At Mercy, we are so grateful we have the systemwide support and reallocation of resources we need to participate in the FDA trials. It's exciting to work together to try to help as many people as we can. We plan to keep pressing forward to expand our program."
Trinity Health Of New England is contributing to convalescent plaza research in COVID-19 treatment
Trinity Health Of New England is participating in a Food and Drug Administration study to assess the safety and efficacy of using convalescent plasma to treat COVID-19.
It is the fourth health care system to receive approval to do the testing as part of a phase two clinical trial. Others include the Mayo Clinic, the Johns Hopkins Health System and the Mount Sinai Health System. Trinity Health Of New England is a member of Trinity Health.
"Convalescent plasma is a new old treatment," says Dr. Danyal Ibrahim, regional chief quality officer for Trinity Health Of New England. "Most recently, it was used in two reports from China. One, published in the Journal of the American Medical Association, spoke of five critically ill individuals in ICU who all showed significant recovery improvement from coronavirus within five days. Another case series used ten individuals, the majority critically ill, who all demonstrated strong evidence of improvement as well."
Those Chinese reports, says Ibrahim, motivated Trinity Health Of New England to approach the FDA to do a clinical trial in its region.
"This is a great opportunity to evaluate results scientifically and to measure risks/benefits," says Ibrahim. If we can demonstrate that convalescent plasma works, it holds great promise because 80 percent of individuals with COVID-19 have mild symptoms and recover well from the disease," says Ibrahim. "That mean we could potentially have a very large pool of donors."
The health system is partnering with a regional blood bank in the research. The study will enroll patients who are in ICU and on ventilators. The intention is to run a 30-day trial and then share results with the FDA. According to WWLP in Hartford, Connecticut, Trinity Health of New England treated its first patient in the study on April 23.
The hope is that if convalescent plasma is demonstrated to be effective, it can then be used on patients with COVID-19 before they require ventilators, and perhaps even prophylactically on frontline healthcare workers.
— RENEE STOVSKY